Which of the Following Best Describes a Double-replacement Reaction
I believe the correct answer from the choices listed above is option B. It is a double replacement because the reaction starts with two compounds and ends with two compounds where the positive and negative ions have changed places.
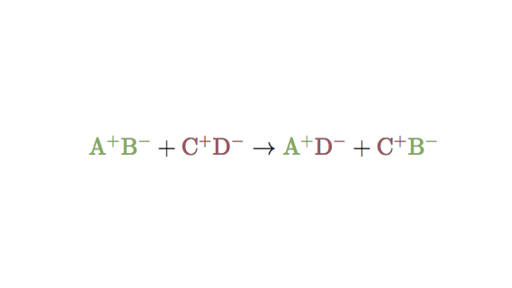
Double Replacement Reactions Double Displacement Article Khan Academy
2AgNO3 Cu 2Ag Cu NO32.
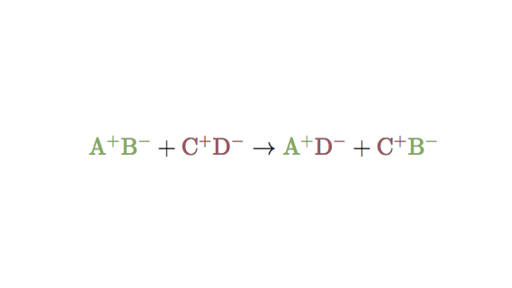
. One compound breaks down into two or more simpler substances. Double replacement reaction O acid base reaction. You can think of the reaction as swapping the cations or the anions but not swapping both since you would end up with the same substances you started with.
Therefore a chemical reaction will occur and silver phosphate and lithium nitrate are the products. Find an answer to your question Which of the following best represents a double replacement reaction. Which of the following demonstrates a double-replacement reaction.
Which of the following best describes the reaction if any that occurs when aqueous solutions of iron III nitrate and sodium iodide are combined. Which of the following best describes a double-replacement reaction. While the Sodium ion on the hydroxide has been replaced by the Hydrogen ion.
Two different compounds exchange positive ions and form two new compounds. A B AB C. Thats why we take the recruitment process seriously to have a.
DESCRIPTION- the reaction is double displacement reactiondouble replacement reactionmetathesis double. 2Mg O2 2MgO. Identify the precipitate formed when solutions of the following ionic compounds are mixed.
A chemical reaction will do the following. See answer 1 Best Answer. According to rule 8 in Table 72 Ag3PO4 is insoluble meeting the requirements for a double-replacement reaction to happen.
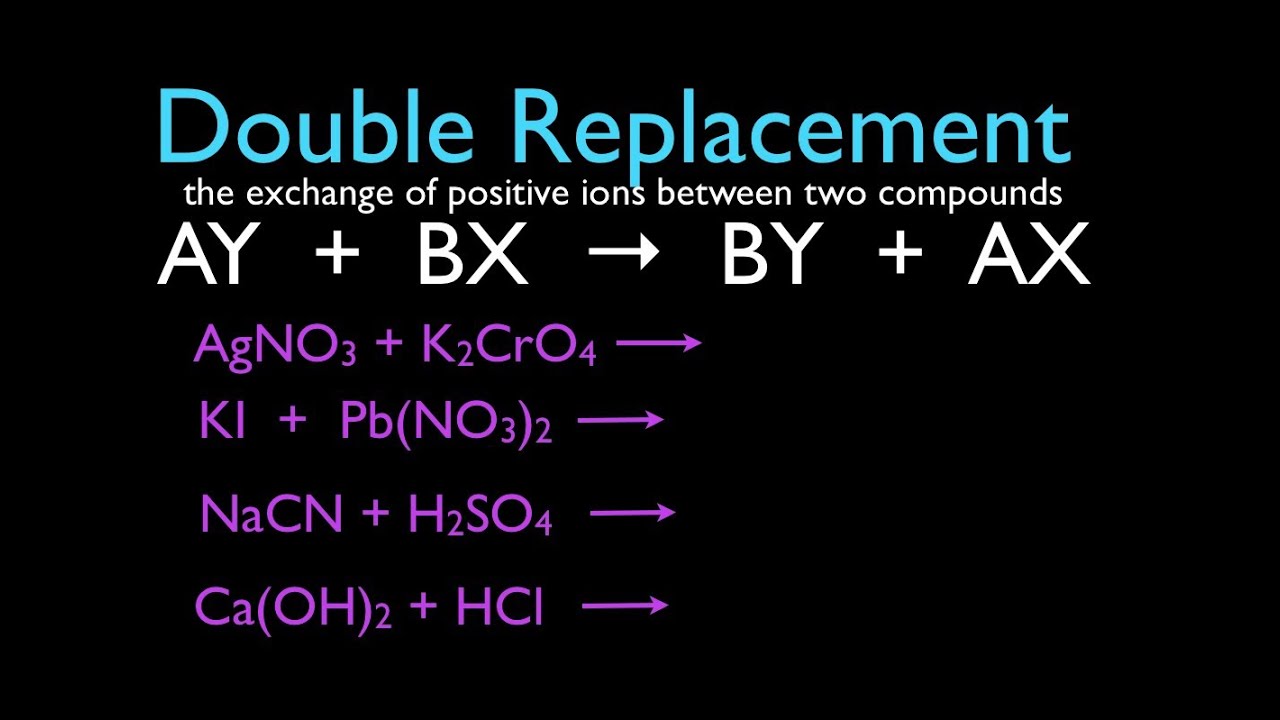
A double-replacement reaction happens when atoms in one compound switch places with atoms in another compound. AB CD AD CB. The general form of a double-replacement also called double-displacement reaction is.
The the following reaction is. Draw curved arrows for the following reaction step. Formation of liquid water neutralization rxn Acid Base H₂O ionic compound.
The positive hydrogen ion on the Chlorine has been replaced by a positive sodium ion on the Chlorine. Which of the following best represents a double replacement reaction. What term refers to a chemical reaction that absorbs heat energy.
CaCO3 CaO CO2. AB A B B. The solvent for a double replacement reaction is usually water and the reactants and products are usually ionic compoundsbut they can also be acids or bases.
Change State Be able to change back to the original materials. Two or more substances react to form a single substance. Types of DR reactions.
Which of these statements best describes a double-replacement reaction. PbNO32 2KI -- PbI2 2KNO3 FeS 2HCl -- FeCl2 H2S. It is a type of chemical reaction where two compounds react and the positive ions cation and the negative ions anion of the two reactants switch places.
The ions of two compounds exchange places to form two new compounds. The ions of two aqueous compounds exchange places to form two new compounds. In this reaction A and C are positively-charged cations while B and D are negatively-charged anions.
Atoms in one compound switch places with atoms in another compound. Chemical Reactions 1 Of 11 Double Replacement Reactions An Explanation Youtube. Terms in this set 2 Double Replacement Reactions.
-Formation of liquid water neutralization rxn -Formation of a precipitate. Double replacement reactions also called double displacement exchange or metathesis reactions occur when parts of two ionic compounds are exchanged making two new compounds. Na3PO4 MnCl2 Mn3 PO42 NaCl.
AX BYAY BX. When the two reactants are mixed the potential products are Ag3PO4 and LiNO3. Gas Ionic molecular compound 3 more precipitate double-displacement reaction double-replacement reaction.
This group of writers have passed strict English tests plus tests from their fields of specialization. AB A B A B AB A BC AC B AC BD AD BC. An element takes the place of another element in a compound.
A BC AC B D. SCICHE515 Double-Replacement Reactions - Chemistry. This means they recently joined the team.
Double replacement reaction O acid base. Write a sentence that completely describes the chemical reaction represented by this balanced equation. When silver nitrate AgNO3aq reacts with hydrochloric acid HClaq a chemical reaction takes placeAgNO3aq HClaq --- AgCls HNO3aqThis is specifically called a double replacement.
Double replacement reactions are also called double replacement reactions double displacement reactions or metathesis reactions. Double-replacement reactions generally occur between substances in aqueous solution. View the full answer.
A double replacement reaction is a type of chemical reaction that occurs when two reactants exchange cations or anions to yield two new products. The double-replacement reaction below results in the formation of the precipitate lead. 2HClaq CaCO3s CO2g CaCl2aq H2Ol.
Change colour Create smoke A non chemical reaction Physical Reaction will do the following.

You Ll Need To Recognize Each Of These Reactions For The Teas Science Section Chemistry Worksheets Chemistry Lessons Teaching Chemistry
Chemical Reactions 1 Of 11 Double Replacement Reactions An Explanation Youtube

Decomposition Reactions Youtube Chemical Reactions Chemical Equation Reactions

Combustion Reactions Chemical Reactions Chemical Equation Reactions
Comments
Post a Comment